Why are natural antioxidants popular in pet nutrition?
An antioxidant is a substance that stops free radicals from causing damage to cells, and to prevent or delay the oxidation of fat or food decomposition and deterioration. There are two main types of antioxidants: synthetic and natural. Synthetic antioxidants are chemically derived and include ethoxyquin, butylated hydroxytoluene, and BHA. Natural antioxidants are generally found in fruit and vegetables and are important for maintaining a healthy immune system and quality of life, including vitamin E, lutein, and beta carotene. Vitamin C is also an effective antioxidant. These ingredients can be added to a dog’s diet or purchased in supplements, they can improve your pet’s immune system and make him more energetic.
During the processing of food, synthetic antioxidants are used to control the chain of reactions involving free radicals. Many food processors are now embracing clean-label antioxidants. These antioxidants help keep the meat, poultry, and fish in a fresh state. However, many people have concerns about the safety of synthetic ingredients. Fortunately, these ingredients are considered safe by the U.S. Food and Drug Administration and are allowed in pet foods at low concentrations. Despite the advantages of synthetic ingredients, some consumers prefer to feed their pets with food that contains natural antioxidants. This is because natural antioxidants have lower toxicity levels. Let’s talk about from the definition, and classification of antioxidants and how it works.
Classification of antioxidants
According to the source of raw materials, there are natural antioxidants and synthetic antioxidants. Synthetic antioxidants are chemically synthesized, such as the common TBHQ, BHA, etc. Natural antioxidants are mainly extracted and purified from natural plants or microorganisms, such as common tea polyphenols, rosemary extract, etc.
According to the mechanism of action, it can be divided into main antioxidants and secondary antioxidants. The main antioxidants interrupt or prevent the chain reaction of free radicals by trapping active free radicals, such as TBHQ, tea polyphenols, etc. Secondary antioxidants are mainly used to prevent metal catalytic oxidation by chelating metal ions, or to regenerate primary antioxidants, such as organic acids, phospholipids, etc.
According to the type of use, it can be divided into water-soluble antioxidants and oil-soluble antioxidants. Among them, water-soluble antioxidants can dissolve in water-based materials, and effectively remove water-soluble free radicals, such as tea polyphenols. Oil-soluble antioxidants can be dissolved in oil-based materials to effectively remove free radicals produced by oxidation of oils, such as rosemary extract.
How do natural antioxidants work?
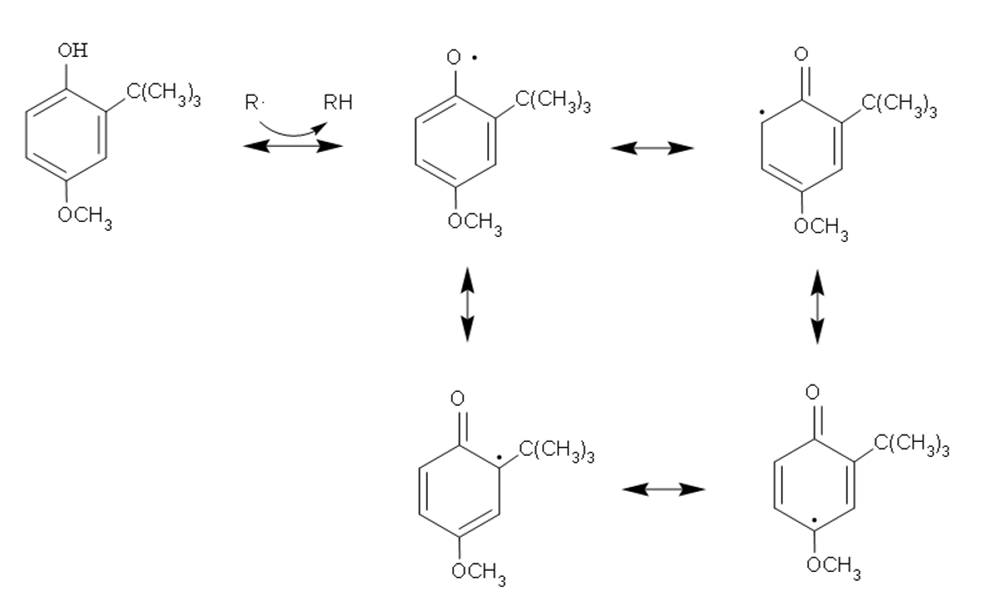
When it comes to lipid oxidation and antioxidant, free radicals cannot be avoided. Free radicals are any atoms or groups that contain one or more unpaired electrons and can exist independently. The food system is catalyzed by certain inducers (such as oxygen, light, enzymes and metal ions) to produce free radicals, which attack fatty acid components and accelerate their oxidation rancidity, resulting in the so-called free radical chain reaction. The effective mechanism of antioxidants is to prevent free radicals, or interrupt free radical chain reactions, and inhibit and slow the automatic oxidation of fats. Phenolic compounds are common structures of natural antioxidants. Their phenolic hydroxyl structure can supply hydrogen to bind free radicals, and enable free radicals to delocalize through the benzene ring structure so that the generated phenolic radical has lower energy and is more stable.
Antioxidant mechanism of phenols
Common natural antioxidants
Natural vitamin E
The most common natural antioxidant in a commercial application, widely distributed in plant tissues, mainly α, β, γ, δ four chemical structures. It has an extremely high safety rating and has been evaluated by the FDA with A safety rating. It is a typical phenolic antioxidant, which can provide hydrogen atoms, bond with free radicals, forms stable intermediate structures or forms new antioxidant components to participate in the reaction and slow down the chain reaction.
Tea polyphenol
The active antioxidant in green tea extract. Stable to heat and acid, with natural, non-toxic and efficient capture of water-soluble free radicals. It is a typical polyhydroxy-phenol antioxidant with a strong hydrogen supply capacity, which can interrupt the chain reaction of hydroperoxide produced by automatic oxidation and form stable compounds to block the oxidation process.
Rosemary Extract
Rosemary is a labiaceae plant, containing carnosic acid, carnosol, rosmarinic acid and other effective ingredients. Rosemary extract has good heat resistance (stable at 200℃), UV resistance, high efficiency, non-toxic antioxidant effect. It is a phenolic antioxidant with the structure of o-phenol, which can not only absorb free radicals but also destroy singlet oxygen and inhibit photosensitive oxidation reactions.
Effects of antioxidants on the shelf life of pet food
Ensure product safety and nutrition
Oxidative rancidity will lead to the oxidation of unsaturated fatty acids in fats into hydroperoxides, and then decompose into small molecules of aldehydes, ketones, acids, etc. Aldehydes and ketones can react with almost all biological components inside and outside the cell and produce toxic effects, resulting in a variety of intracellular and extracellular damage. Lipid oxidation can cause nutrient reduction in pet food. According to studies, intake of oxidized oil can make animals liver, spleen, pancreas, kidney and other internal organs lesions and enlargement, serious internal tissue degradation atrophy, necrosis. At the same time, essential fatty acids such as linoleic acid and linolenic acid in pet food will be destroyed when oil rancidity occurs, leading to changes in the composition of fatty acids. In addition, the formation of highly active free radicals in the process of oil oxidation can destroy vitamins, especially fat-soluble vitamins such as vitamin A, vitamin E, vitamin D and so on, leading to vitamin deficiency in pets.
Stable product color
Color is an important indicator of product quality in some pet foods. Studies have shown that when the fat components in pet food are oxidized and rancorous, the food body or additional pigment components will also be destroyed. However, studies have found that, in the face of pet food containing certain pigment components, adding an appropriate amount of natural antioxidants in the production process of the product can effectively prevent or delay the oxidation reaction, stabilize the sensory properties of the product, and thus extend the shelf life of the product. And in the actual shelf life process, consumers are advised to store pet food away from light to reduce the damage of light to pigment components.
Ensure the food palatability
For fat-rich pet food, in the process of processing and shelf life, fat oxidation will produce secondary oxidation products such as aldehydes, acids, ketones and other volatile substances, causing pet food to produce an irritating odor when the accumulated concentration is high enough, may seriously affect the palatability of pet food.